This standard defines the requirements for a quality management system as to development, manufacturing, storage and distribution, installation, maintenance, final taking out of operation and disposal of medical devices. General information on the ISO 13485:2016 Yet, this article will focus on the risk-based approach for business process control according to ISO 13485. There are different ways to address this concept. However, its last revision in 2016 introduced an interesting approach that is still late-breaking: The risk-based approach as a part of quality management. 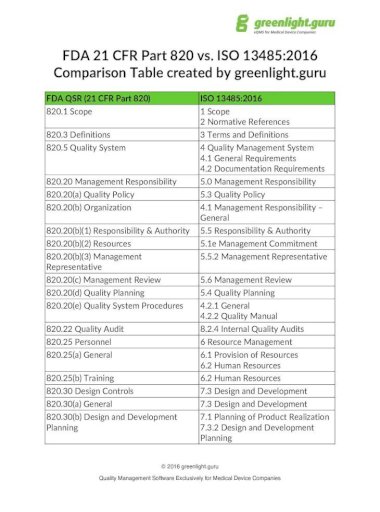
It places requirements on a quality management system for all stages of a medical device life cycle. The standard ISO 13485 is widely known among medical device manufacturers, being an established standard.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
